You are using an outdated browser. Please
upgrade your browser to improve your experience.
TrialKit
TrialKit
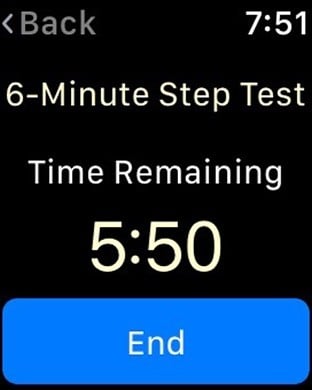
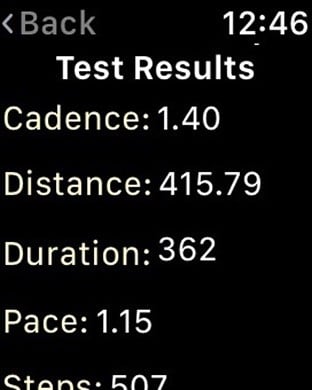
Leverage the latest in data collection and study management technology to expedite clinical trials and advance healthcare. TrialKit eliminates the need for costly, outdated data collection tools by embracing the accessibility and efficiency of mobile operating systems.The mobility and versatility of TrialKit allow for seamless regulatory compliant (21 CFR Part 11) data capture from mobile devices anytime, anywhere. Once collected, data can be easily aggregated, analyzed and shared, making collaboration amongst research teams more productive. Best of all, no programming expertise is needed to use TrialKit, so any research professional can create a study using an iOS device. How does it work?1. Build a studyAn intuitive user interface enables study builders to create electronic case report forms (eCRFs) to collect essential study data in accordance with the study protocol and regulatory requirements. Ensure accurate data collection with easy-to-create edit checks. 2. Collect dataTrialKit enables research teams to collect and cleanse clinical data from virtually any location. Export data in a variety of formats for review and submission and capture ePRO data with ease.3. Manage workflowWith TrialKit, clinical research can be expertly managed from start to finish on an iPhone or iPad. Configuring role-based security, form review levels, and risk-based monitoring is simple. The ability to access, monitor, and review data or respond to queries on-the-go is now a reality. 4. Engage study participantsThe ePRO capabilities built into TrialKit will enhance your clinical trials. Automate the entire notification process and enforce patient survey completion by sending reminders. Study participants’ access has been made simple and flexible, so little or no training is required. Email and/or in-app notifications guide patients directly to their surveys, which can be accessed through the app or through any web browser.5. Analyze dataRobust reporting options enhance the ways research teams view and analyze data gathered in clinical trials. In addition to My Queries, Action Items, Regulatory Audit, and Outcome Summary reports, TrialKit generates a dynamic, visual report to track study progress in real-time.Unique Features and Functionality:Device-targeted eCRF designImage and video capture directly to eCRFsSite and study document supportBuilt-in ePRO, including a patient handoff modeAdjudication for endpoints, outcomes, and inclusion/exclusion criteriaInventory management - Manage both drug and device inventory with a device you carry in your pocket. Barcode scanning eliminates human error and gives you real-time drug and or device dispositionHarness the Power of iOS - Imagine uploading a photo, movie or voice narration to eCRF instantly and into the database. Program push notifications. Stamp GPS location to any record and view on a map. Operate in both landscape and portrait mode on any iOS device from an iPhone 5 on up. Sign case report forms with your finger or stylus. Activity Testing - Patients can now truly participate in their own treatments. Activity testing allows research professionals to measure a variety of different types of activity, using Core Motion and HealthKit. Drop range of motion, step motion, or heart rate tests directly into any visit interval within any study To learn more about this technology that allows the future of clinical trials to be possible today, visit https://www.crucialdatasolutions.com/.
Features